Riluzole
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Rilutek, Tiglutik, Exservan, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a696013 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 60±18%[4] |
| Protein binding | 97%[4] |
| Metabolism | Hepatic (CYP1A2)[4] |
| Elimination half-life | 9–15 hours[4] |
| Excretion | Urine (90%)[4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.124.754 |
| Chemical and physical data | |
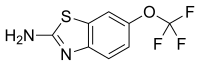
| Formula | C8H5F3N2OS |
| Molar mass | 234.20 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Riluzole is a medication used to treat amyotrophic lateral sclerosis (ALS) and other motor neuron diseases. Riluzole delays the onset of ventilator-dependence or tracheostomy in some people and may increase survival by two to three months.[5] Riluzole is available in tablet and liquid form.
Medical use
[edit]Amyotrophic lateral sclerosis
[edit]Riluzole was approved in the United States for the treatment of ALS by the U.S. Food and Drug Administration (FDA) in 1995.[6] A Cochrane Library review states a 9% gain in the probability of surviving one year.[5]
Adverse effects
[edit]- Very common (>10% frequency):[7] nausea; weakness; decreased lung function
- Common (1–10% frequency):[8] headache; dizziness; drowsiness; vomiting; abdominal pain; increased aminotransferases
- Uncommon (0.1–1% frequency):[8] pancreatitis; interstitial lung disease
- Rare (<0.1% frequency):[8] neutropenia; allergic reaction (including angiooedema, anaphylactoid reaction)
Overdose
[edit]Symptoms of overdose include: neurological and psychiatric symptoms, acute toxic encephalopathy with stupor, coma and methemoglobinemia.[4] Severe methemoglobinemia may be rapidly reversible after treatment with methylene blue.[4]
Contraindications
[edit]Contraindications for riluzole include: known prior hypersensitivity to riluzole or any of the excipients inside the preparations, liver disease, pregnancy or lactation.[4]
Interactions
[edit]CYP1A2 substrates, inhibitors and inducers would probably interact with riluzole, due its dependency on this cytochrome for metabolism.[4]
Mechanism of action
[edit]Riluzole preferentially blocks TTX-sensitive sodium channels, which are associated with damaged neurons.[9][10] Riluzole has also been reported to directly inhibit the kainate and NMDA receptors.[11] The drug has also been shown to postsynaptically potentiate GABAA receptors via an allosteric binding site.[12] However, the action of riluzole on glutamate receptors has been controversial, as no binding of the drug to any known sites has been shown for them.[13][14] In addition, as its antiglutamatergic action is still detectable in the presence of sodium channel blockers, it is also uncertain whether or not it acts via this way. Rather, its ability to stimulate glutamate uptake seems to mediate many of its effects.[15][16] In addition to its role in accelerating glutamate clearance from the synapse, riluzole may also prevent glutamate release from presynaptic terminals.[17] Since CK1δ plays a key role in TDP-43 proteinopathy, a pathological hallmark of ALS, this could help to better decipher drug mechanism of action.
Synthesis
[edit]Riluzole can be prepared beginning with the reaction of 4-(trifluoromethoxy)aniline with potassium thiocyanate followed by reaction with bromine, forming the thiazole ring.[18][19][20]
Society and culture
[edit]Legal status
[edit]Riluzole was approved for medical use in the European Union in October 1996.[3]
Research
[edit]A number of case studies and randomized controlled trials have indicated that riluzole, which is neuroprotective and a glutamate modulator, may have use in mood and anxiety disorders.[21][22][23] However, it failed in trials of Huntington's disease and Parkinson's disease.[23]
Riluzole has been investigated in rodent models for its potential ability to protect against noise-induced hearing loss (NIHL) and cisplatin-induced ototoxicity. These protective effects are believed to be caused by riluzole's antioxidant and anti-apoptotic properties, but other mechanisms, including modulation of glutamate signaling, are also being investigated.[24][25] However, further research, especially in human trials, is necessary to confirm these findings and establish riluzole's clinical efficacy for treating hearing loss.
A sublingual reformulation of riluzole that originated at Yale University and is known by the code name BHV-0223[26] is under development[when?] for the treatment of generalized anxiety disorder and mood disorders by Biohaven Pharmaceuticals.[27][28] A prodrug formulation of riluzole, troriluzole, has been researched as a potential treatment for several different conditions.[29][30]
See also
[edit]References
[edit]- ^ "Rilutek (riluzole) tablets, for oral useInitial U.S. Approval: 1995". DailyMed. Retrieved August 26, 2024.
- ^ https://www.accessdata.fda.gov/drugsatfda_docs/nda/pre96/020599Orig1s000rev.pdf [bare URL PDF]
- ^ a b "Rilutek EPAR". European Medicines Agency (EMA). April 16, 2007. Retrieved October 1, 2020.
- ^ a b c d e f g h i "PRODUCT INFORMATION RILUTEK® (riluzole) Tablets" (PDF). TGA eBusiness Services. sanofi-aventis australia pty ltd. January 6, 2009. Retrieved February 18, 2014.
- ^ a b Miller RG, Mitchell JD, Moore DH (March 2012). "Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND)". The Cochrane Database of Systematic Reviews. 2012 (3): CD001447. doi:10.1002/14651858.CD001447.pub3. PMC 7055506. PMID 22419278.
- ^ "Riluzole". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases. May 2018. PMID 31644225.
- ^ "Rilutek (riluzole) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved February 18, 2014.
- ^ a b c Rossi S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- ^ Song JH, Huang CS, Nagata K, Yeh JZ, Narahashi T (August 1997). "Differential action of riluzole on tetrodotoxin-sensitive and tetrodotoxin-resistant sodium channels" (PDF). The Journal of Pharmacology and Experimental Therapeutics. 282 (2): 707–714. PMID 9262334.
- ^ Bellingham MC (February 2011). "A review of the neural mechanisms of action and clinical efficiency of riluzole in treating amyotrophic lateral sclerosis: what have we learned in the last decade?". CNS Neuroscience & Therapeutics. 17 (1): 4–31. doi:10.1111/j.1755-5949.2009.00116.x. PMC 6493865. PMID 20236142.
- ^ Debono MW, Le Guern J, Canton T, Doble A, Pradier L (April 1993). "Inhibition by riluzole of electrophysiological responses mediated by rat kainate and NMDA receptors expressed in Xenopus oocytes". European Journal of Pharmacology. 235 (2–3): 283–289. doi:10.1016/0014-2999(93)90147-a. PMID 7685290.
- ^ He Y, Benz A, Fu T, Wang M, Covey DF, Zorumski CF, et al. (February 2002). "Neuroprotective agent riluzole potentiates postsynaptic GABA(A) receptor function". Neuropharmacology. 42 (2): 199–209. doi:10.1016/s0028-3908(01)00175-7. PMID 11804616. S2CID 24194421.
- ^ Wokke J (September 1996). "Riluzole". Lancet. 348 (9030): 795–799. doi:10.1016/S0140-6736(96)03181-9. PMID 8813989. S2CID 208788906.
- ^ Kretschmer BD, Kratzer U, Schmidt WJ (August 1998). "Riluzole, a glutamate release inhibitor, and motor behavior". Naunyn-Schmiedeberg's Archives of Pharmacology. 358 (2): 181–190. doi:10.1007/pl00005241. PMID 9750003. S2CID 5887788.
- ^ Azbill RD, Mu X, Springer JE (July 2000). "Riluzole increases high-affinity glutamate uptake in rat spinal cord synaptosomes". Brain Research. 871 (2): 175–180. doi:10.1016/S0006-8993(00)02430-6. PMID 10899284. S2CID 23849619.
- ^ Dunlop J, Beal McIlvain H, She Y, Howland DS (March 2003). "Impaired spinal cord glutamate transport capacity and reduced sensitivity to riluzole in a transgenic superoxide dismutase mutant rat model of amyotrophic lateral sclerosis". The Journal of Neuroscience. 23 (5): 1688–1696. doi:10.1523/JNEUROSCI.23-05-01688.2003. PMC 6741992. PMID 12629173.
- ^ Wang SJ, Wang KY, Wang WC (January 2004). "Mechanisms underlying the riluzole inhibition of glutamate release from rat cerebral cortex nerve terminals (synaptosomes)". Neuroscience. 125 (1): 191–201. doi:10.1016/j.neuroscience.2004.01.019. PMID 15051158. S2CID 35667296.
- ^ Yagupol'skii LM, Gandel'sman LZ (1963). "Missing". Zh. Obshch. Khim. 33: 2301.
- ^ EP 50551, Mizoule J, "Medicament containing 2-amino-6-trifluoro-methoxy benzothiazole", published 1982-04-28, assigned to Pharmindustrie; US 4370338, Mizoule J, issued 1983, assigned to Pharmindustrie
- ^ U.S. patent 4,826,860
- ^ Kawashima Y, Yamada M, Furuie H, Kuniishi H, Akagi K, Kawashima T, et al. (September 2023). "Effects of riluzole on psychiatric disorders with anxiety or fear as primary symptoms: A systematic review". Neuropsychopharmacology Reports. 43 (3): 320–327. doi:10.1002/npr2.12364. PMC 10496048. PMID 37463744.
- ^ Grant P, Song JY, Swedo SE (August 2010). "Review of the use of the glutamate antagonist riluzole in psychiatric disorders and a description of recent use in childhood obsessive-compulsive disorder". Journal of Child and Adolescent Psychopharmacology. 20 (4): 309–315. doi:10.1089/cap.2010.0009. PMC 2958461. PMID 20807069.
- ^ a b Zarate CA, Manji HK (September 2008). "Riluzole in psychiatry: a systematic review of the literature". Expert Opinion on Drug Metabolism & Toxicology. 4 (9): 1223–1234. doi:10.1517/17425255.4.9.1223. PMC 2587133. PMID 18721116.
- ^ Üstün Bezgin S, Uygur KK, Gökdoğan Ç, Elmas Ç, Göktaş G (July 2019). "The Effects of Riluzole on Cisplatin-induced Ototoxicity". International Archives of Otorhinolaryngology. 23 (3): e267–e275. doi:10.1055/s-0038-1676654. PMC 6660296. PMID 31360245.
- ^ Ruel J, Wang J, Pujol R, Hameg A, Dib M, Puel JL (July 2005). "Neuroprotective effect of riluzole in acute noise-induced hearing loss". NeuroReport. 16 (10): 1087–90. doi:10.1097/00001756-200507130-00011. PMID 15973153. S2CID 29393000.
- ^ "BHV 0223 – AdisInsight". Adisinsight.springer.com. Retrieved May 20, 2016.
- ^ Harris E (2015). "Industry update: the latest developments in therapeutic delivery". Therapeutic Delivery. 6 (6): 647–652. doi:10.4155/tde.15.44. ISSN 2041-5990.
- ^ Terry M (August 16, 2018). "Biohaven Reports Positive Early Clinical Trial Results for Anxiety Drug". BioSpace. Retrieved November 5, 2023.
- ^ van Roessel PJ, Grassi G, Aboujaoude EN, Menchón JM, Van Ameringen M, Rodríguez CI (January 2023). "Treatment-resistant OCD: Pharmacotherapies in adults". Comprehensive Psychiatry. 120: 152352. doi:10.1016/j.comppsych.2022.152352. hdl:2445/192315. PMID 36368186.
- ^ Silk AW, Saraiya B, Groisberg R, Chan N, Spencer K, Girda E, et al. (July 2022). "A phase Ib dose-escalation study of troriluzole (BHV-4157), an oral glutamatergic signaling modulator, in combination with nivolumab in patients with advanced solid tumors". European Journal of Medical Research. 27 (1): 107. doi:10.1186/s40001-022-00732-w. PMC 9250196. PMID 35780243.

